
Therefore, it needs two more electrons to become stable (achieve octet electronic configuration). Moving on, the Oxygen atom ( O 8) has six electrons in its outer shell. The Hydrogen atom follows the duplet rule that says: An atom is stable if it has two electrons in its outer shell. 
To become stable, the hydrogen atom needs one electron to achieve a duplet electronic configuration (the duplet rule). If I talk about hydrogen ( H 1), it has one electron in its outermost shell. If I talk about water (H2O), we will be talking about the first method. The sharing of electrons between atoms of the same elements (for example N2, H2 and O2 etc).The sharing of electrons between atoms of different elements (for example H2O, CH4 and NH3 etc).The two ways in which covalent bonding can be achieved: Plus, the elements having a low electron affinity cannot take up electrons.Īs a result, the atoms of such elements share electrons with each other (to achieve stability). The answer is simple, ionisation energy (energy essential to take away an electron).Įlements that have high ionisation energy cannot transfer electrons. Important Note: At this stage, you might be wondering, why not atoms exchange electrons like ionic bonding? Why does this exchange of electrons take place? So, whenever an atom becomes stable, it is said to have achieved the electronic configuration of a noble gas. Noble gases are unreactive (as they have fully-filled outer shells). Teachers can devote more time to coaching their students, helping them develop procedural fluency (if needed), and assisting them with challenging projects that allow them to exert greater control over their own learning (Hamdan, McKnight, McKnight & Arfstrom, 2013b).In an outermost shell, an atom should have two or eight electrons to achieve the atomic configuration of a noble gas. Time becomes available for students to collaborate with peers on projects, engage more deeply with content, practice skills, and receive feedback on their progress. Transforming direct instruction into a self-directed learning approach allows teachers to reconsider how to maximize individual face-to-face time with students. They are, increasingly, turning to an alternative model of instruction termed Flipped Learning in which digital technologies are used to shift direct instruction outside of the group learning space to the individual learning space, usually via videos. There is not much room to differentiate instruction based on students’ interests, learning styles, and readiness, which are critical elements to consider in attempting to address the diverse needs of students (Tomlinson, 2014).Įducators have been working to break this lecture-centered instructional model by shifting the focus from the curriculum-driven instruction to student learning needs to drive instruction. Teachers’ instruction is largely driven by the amount of material to cover before the test at the end of the unit. Teachers make their best effort to keep the class on schedule. Students’ additional questions are more likely to be left unanswered due to the lack of class time. The next day, the teacher will collect and briefly review the homework assignment. The teacher may be acutely aware that many students do not understand the day’s lesson, but does not have the time to meet with them individually during the 50-minute class period. In assuming a micro perspective of teaching, educators’ efforts to move beyond lower levels of Bloom’s Taxonomy can be difficult within limited class-time constraints. Students are more likely to locate information for their own learning purposes through this pedagogical framework. 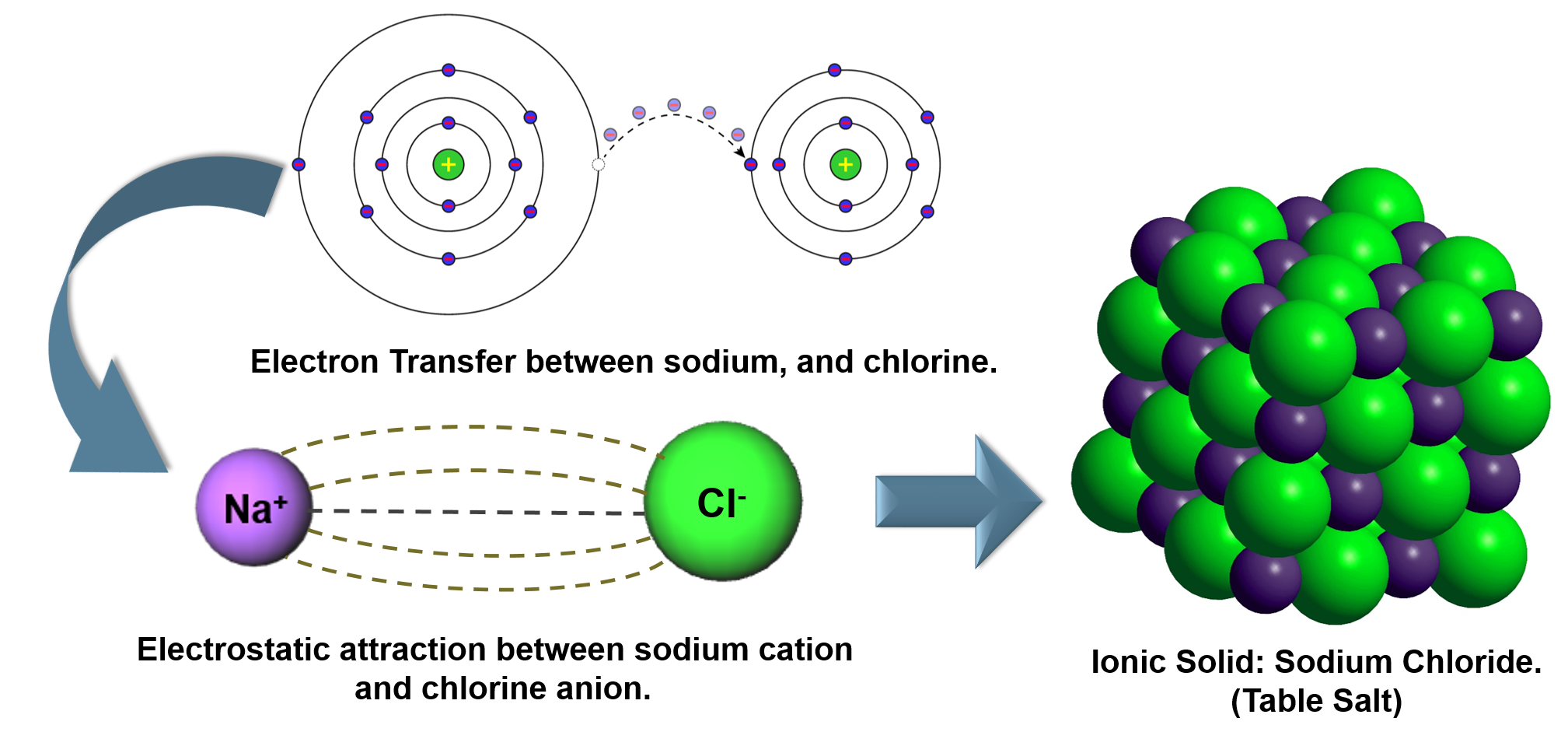
Additionally, direct teaching lecture methods fail to consider students’ self-directed learning tendencies. 
Students have become accustomed to learning with digital devices and multimedia. Direct teaching methods consisting of lecture and auditing are not well aligned with the ways modern students improve their literacy skills. Traditional norms of direct teaching methods have faced various challenges to meet students’ needs in advancing knowledge to become 21 st century global competitors. It will describe how a flipped classroom model approach can help educators meet long-standing challenges while supporting teachers in meeting the diverse needs of their students. This first section will highlight current challenges in education.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
